by UKCDR & GloPID-R
Research funders recognise that there is a need to coordinate COVID-19 research funding at all levels to prevent duplication and improve impact. This coordination should not be limited to the COVID-19 pandemic, but instead needs to facilitate ongoing improved coordination for future infectious disease epidemics and pandemics.
UKCDR and the Global Research Collaboration for Infectious Disease Preparedness (GloPID-R) have agreed a set of principles to align research funders towards a coordinated effort for supporting high-quality research for the most pressing global needs in epidemics and pandemics.
Funders who have committed to the principles so far include:

FUNDER PRINCIPLES FOR SUPPORTING HIGH-QUALITY RESEARCH FOR THE MOST PRESSING GLOBAL NEEDS IN EPIDEMICS & PANDEMICS
These principles are proposed for endorsement by research funders, donors, governments or any other entities supporting research to address the most pressing global needs on COVID-19 and for future epidemics and pandemics (collectively referred as “the funders”). The core principles are intended to be applicable for any epidemic and additional points of relevance for COVID-19 are indicated with an asterisk.
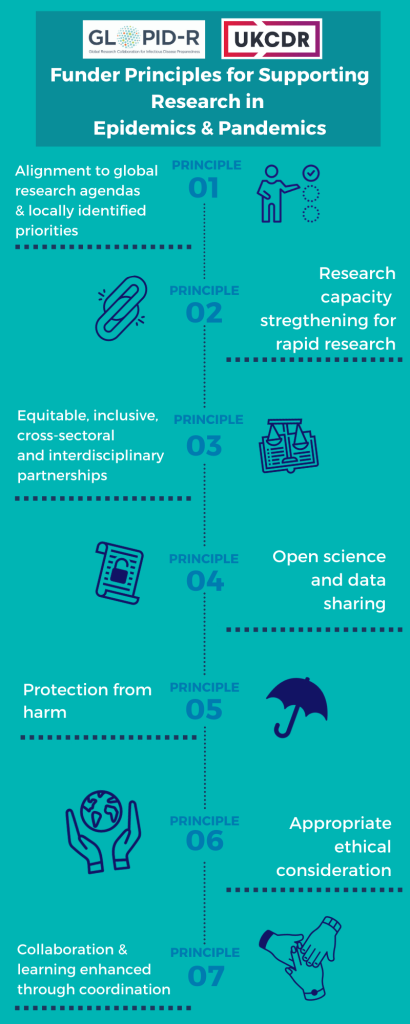
1. Alignment to global research agendas and locally identified priorities
To consider global research priorities, such as proposed by the World Health Organisation (WHO) and other multilateral entities or regional bodies such as the African Union, as well as local research priorities, in addition to funder strategic priorities, when funding research for global benefit.
The WHO R&D Blueprint was developed to help guide the research response for epidemics and pandemics and alignment with this and associated research roadmaps developed for a coordinated response focusses the funds available. It is recognised that certain global research priorities (or additional priorities) may be of particular relevance for research in resource limited settings and consideration of locally identified priorities should also be reflected in the funding process.
* For COVID-19 the WHO Research Roadmap for COVID-19 has been developed by the WHO R&D Blueprint team building on consensus from global researchers to help guide the research response for COVID-19*
2. Research capacity for rapid research
a. To build upon existing research capacity and systems, where available.
For research to inform the health, economic and social policy and public health response in an ongoing epidemic or pandemic (or future outbreaks of the same pathogen), it needs to be implemented as rapidly as possible. Funders recognise that building on existing research capacity and systems is the fastest way to ensure high quality research is conducted and knowledge exchanged and that the long-term impacts of epidemics and recovery are addressed. Incorporation of epidemic relevant research questions into existing research studies (for example cohorts and clinical research networks) will be encouraged where possible, applicable and appropriate, to gain benefits from both rapid research activation, knowledge mobilisation and pre-existing relevant data.
b. To support capacity strengthening necessary for the research.
Funders recognise the need for strengthening research capacity in particular in resource limited settings and will consider the sustainability of any newly funded research capacity and whether it could be embedded for rapid activation in future outbreaks. Relevant guidance is provided by the work of the ESSENCE Group including the ESSENCE Good Practice Document on Capacity Strengthening.
3. Equitable, inclusive, cross-sectoral and interdisciplinary partnerships
a. To support equitable partnership throughout the research process.
Equitable partnerships are needed to ensure successful, embedded research, which is locally relevant. Partnerships supported should be informed by relevant guidance such as UKCDR’s Equitable Partnership Principles; COHRED’s Research Fairness Initiative and Fair Research Contracting, and the Commission for Research Partnerships with Developing Countries (KFPE) 11 Principles for Research Partnership.
* Funders may additionally support the aspiration that any new vaccines, diagnostics, and treatments developed for COVID-19 are globally available, appropriate, and affordable, regardless of where they have been developed or who has funded them, aligned with the Global Collaboration ACT Accelerator. *
b. To promote inclusive and cross-sectoral partnerships to ensure that research is most likely to impact policy and practice.
Inclusivity is needed to ensure consideration of vulnerable or marginalised groups in the research agenda. Public and community engagement plays a particularly important role in achieving and maintaining trust for research within communities for research during outbreaks, informed by guidelines such as the UNAIDS Good Participatory Practice Guidelines for Biomedical HIV prevention Trials. Research partnerships should demonstrate that community and public engagement has taken place and will continue to do so.
Cross-sectoral partnerships across communities, government, public health and non-governmental organisations all help to ensure that the research funded is most likely to impact policy and practice for the relevant government and public health organisations.
c. To promote interdisciplinary research
The importance of interdisciplinary partnerships for relevant and effective research in epidemics has been highlighted, including through the joint work of the UK Academy of Medical Sciences, UK Medical Research Council and InterAcademy Partnership.
4. Open science and data sharing
To require that research findings and data relevant to the epidemic are shared rapidly and openly to inform the public health response.
Rapid research findings, data sharing and open access publishing can accelerate health benefits through; facilitating research projects; reducing the duplication of work; and ensuring a clearer picture of the disease through pooled results to improve intervention effectiveness. Funders will be informed by relevant guidance such as the GloPID-R Roadmap for Data Sharing (in particular, the guidance on grant conditions requiring rapid sharing of quality assured data and development and review of data management plans in alignment with the FAIR Guiding Principles for scientific data management and stewardship) as well as the associated GloPID-R Principles of Data Sharing in Public Health Emergencies (Timely, Ethical, Accessible, Transparent, Equitable, Fair, Quality).
* For COVID-19 the joint statement on Sharing research data and findings relevant to the novel coronavirus (COVID-19) outbreak is pertinent*
5. Protection from harm
To take all reasonable steps to anticipate, mitigate and address harm to those involved with research funded.
Everyone involved in the research chain, from research funders, planners and practitioners to local community members, has the right to be safe from harm. Funders working in international development research will be informed by guidance such as UKCDR’s guidance on safeguarding in international development research.
* For COVID-19 there is a companion piece on practical application of the UKCDR safeguarding guidance during COVID-19.*
6. Appropriate ethical consideration
To ensure appropriate ethical consideration is embedded throughout research conducted, in particular regarding access to the products of research.
Ethics should be at the heart of funding decision-making and considered throughout the research, including informing approaches to ensure that the optimal value is being obtained from the research for all parties involved. Relevant guidance is provided by the Declaration of Helsinki International Ethical Guidelines for Health-related research involving humans by the Council for International Organizations of Medical Sciences (CIOMS) , Nuffield Bioethics for public health emergencies – recommendations and The Global Code of Conduct for Research in Resource-Poor Settings.
* For COVID-19 the WHO Ethical Standards for research During Public Health emergencies: Distilling Existing Guidance to Support COVID-19 R&D is pertinent. *
7. Collaboration and learning enhanced through coordination
Coordination to ensure maximum impact of investments for research on the most pressing global needs for epidemics through cross- funder and cross- researcher collaboration learning and evaluation.
a. To map research funded, use these data to enhance coordination, and ensure it is publicly available.
Maximising the value of research investments requires accessible, comprehensive and coherent information on what and where others are investing to help identify funding gaps or duplication and inform or direct future investments. Research funded needs to be mapped publicly, for example through World Report.
*For COVID-19 the COVID-19 Research Project Tracker by UKCDR & GLOPID-R is pertinent. The Research Project Tracker is aligned with the WHO Research Roadmap for COVID19 to facilitate informed decision making and targeting of funds where there is need.*
b. To foster collaboration between studies funded in epidemics and facilitate shared development of research protocols, data collection tools, data sharing and exchange of knowledge.
Collaboration between researcher communities can facilitate trust, foster new partnerships and improve research outcomes and their impact. Where relevant, funded researchers will be supported to embed in relevant or, co-create communities of practice or an equivalent that promote shared development of research protocols, data collection, purpose driven data and results sharing.
c. To embed operational research where relevant and support impact evaluation across funded projects to learn from and improve future funder and researcher responses for epidemics.
Conducting research during epidemics is still a relatively new endeavour and it is important to embed operational research (research on research) and impact evaluation where relevant. In particular, this should aim to identify how the research response can be improved, including how to overcome barriers to achieving the Funder Principles outlined here (building on prior work undertaken by GloPID-R and GOARN Research such as the PEARLES review and GloPID-R Roadmap for Data Sharing).
We would like to encourage research funders across the globe to join with us and commit to these principles.
SIGN UP TO THE PRINCIPLES HERE